 Order Now
Order Now
Building your website just got easier.

SERVICE
ABOUT CREATIVE CYBER SKY
Creative Cyber Sky is a leading software company in the Pakistan. Our team of highly skilled developers and programmers provide the best solutions to your business needs.
Creative Cyber Sky experienced in website development, CCTV camera installation, attendance machine, web hosting and I.T Consultancy. We have been serving clients since 2015 with our dedication to providing quality services at an affordable price.
We take pride in delivering high quality services to our clients as we understand that you need us more than we need you. Our clients are our top priority and we work hard to give them what they want and need from us.

WHY CHOOSE US
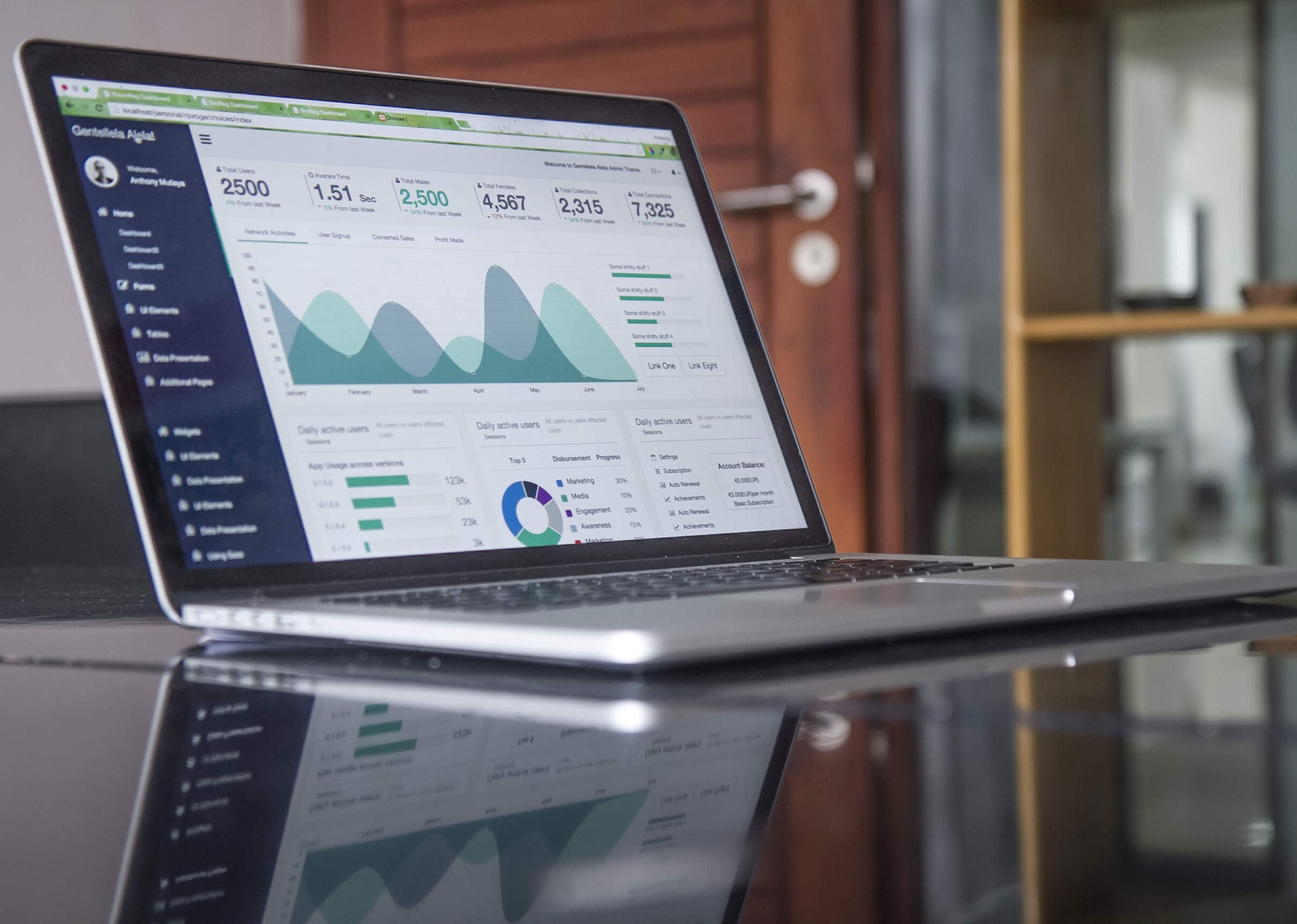
WHAT WE OFFER
POINT OF SALE SYSTEM

Our Tech Stack








Ready to Grow
Social Media
Management
When you are looking for a professional social media handler, you need someone who is going to be able to handle the entire campaign. That means creating a strategy, managing your Instagram and Facebook accounts, taking care of content updates, and much more.
OUR CLINETS









COME & VISIT US
We are located at Pakistan

CUSTOMER CARE
We’re here to help our customers succeed online, and that’s something we hear back from our customers. They just love our support channels.

CUSTOMIZE SOLUTION
If the client's business requires something other than standard SW solutions, a non-traditional procedure or a custom system can be developed.

